Unfortunately it's a combination of the molfile having implicit
stereochemistry (i.e. tools have to guess from the depiction) and the CDK
layout. I can fix both but as a simple work around go through the bonds and
those that are double, not in a ring, or have stereo (check
IStereoElements) should have the stereoType set to E_OR_Z... can't remember
the exact flag.
Will fix though for next release.
J
Regards,
John W May
[email protected]
On 16 January 2015 at 10:05, Oscar Mora <[email protected]> wrote:
> Hi,
>
> I have the following question about processing SMILES format structures +
> 2D Generation + serialization in Mol format. I am using CDK 1.5.8.
>
> My goal is to process an structure in SMILES format, generate 2D
> coordinates and save it as in Mol format. The problem where I am stuck is
> that the input SMILES has the no stereochemistry defined, but the final mol
> output _has_ the stereochemestry defined.
>
> Following I have prepared a simple example to describe my problem:
>
> The input structure is the following one:
>
> ON=CC1=CC=C(O)C=C1
>
> The structure is read and parsed using the following code (snippet):
>
> [...]
> IAtomContainer iAtomContainer = null;
> final SmilesParser smilesParser = new
> SmilesParser(DefaultChemObjectBuilder.getInstance());
> iAtomContainer = smilesParser.parseSmiles(molecule);
>
> [...]
>
> then the 2D coordinates are generated.
>
> [...]
> sdg.setMolecule(iAtomContainer, true);
> sdg.generateCoordinates();
> iAtomContainerWCoordinates = sdg.getMolecule();
> [...]
>
> and finally the 'IAtomContainer' object is serialized as 'mol' file,
> producing the following output:
>
> CDK 0116150922
>
> 10 10 0 0 0 0 0 0 0 0999 V2000
> 3.9132 3.7394 0.0000 O 0 0 0 0 0 0 0 0 0 0 0 0
> 2.6112 2.9947 0.0000 N 0 0 0 0 0 0 0 0 0 0 0 0
> 2.6051 1.4947 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> 1.3030 0.7500 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> 1.3030 -0.7500 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> 0.0280 -1.5000 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> -1.3220 -0.7500 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> -2.6148 -1.5107 0.0000 O 0 0 0 0 0 0 0 0 0 0 0 0
> -1.3220 0.7500 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> 0.0280 1.5000 0.0000 C 0 0 0 0 0 0 0 0 0 0 0 0
> 1 2 1 0 0 0 0
> 2 3 2 0 0 0 0
> 3 4 1 0 0 0 0
> 4 5 2 0 0 0 0
> 5 6 1 0 0 0 0
> 6 7 2 0 0 0 0
> 7 8 1 0 0 0 0
> 7 9 1 0 0 0 0
> 9 10 2 0 0 0 0
> 4 10 1 0 0 0 0
> M END
>
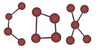
> If you render the structure before (SMILES) and after (MOL) the process,
> it is possible to watch how initially the double bond between N and C has
> not the stereo defined, but at the end of the process is arbitrarily
> defined (E). I have also attached two snapshots two further describe this
> (SMILES_before.png) and (MOL_after.png).
>
> Why is this happening ? Is there any way to control, and keep the flag of
> stereochemistry not defined during the transformation from SMILES to MOL?
> (I would like that the two depictions produce the same image)
>
> Thanks in advance
>
> Oscar
>
>
> ------------------------------------------------------------------------------
> New Year. New Location. New Benefits. New Data Center in Ashburn, VA.
> GigeNET is offering a free month of service with a new server in Ashburn.
> Choose from 2 high performing configs, both with 100TB of bandwidth.
> Higher redundancy.Lower latency.Increased capacity.Completely compliant.
> http://p.sf.net/sfu/gigenet
> _______________________________________________
> Cdk-user mailing list
> [email protected]
> https://lists.sourceforge.net/lists/listinfo/cdk-user
>
>
------------------------------------------------------------------------------
New Year. New Location. New Benefits. New Data Center in Ashburn, VA.
GigeNET is offering a free month of service with a new server in Ashburn.
Choose from 2 high performing configs, both with 100TB of bandwidth.
Higher redundancy.Lower latency.Increased capacity.Completely compliant.
http://p.sf.net/sfu/gigenet
_______________________________________________
Cdk-user mailing list
[email protected]
https://lists.sourceforge.net/lists/listinfo/cdk-user